Pijler Oncologie
De pijler Oncologie van de NVOG heeft de volgende doelen:
- bevorderen van een optimale, integrale zorg voor patiënten met gynaecologische (pre)maligne aandoeningen
- bevorderen van onderwijs en onderzoek op het gebied van de oncologische gynaecologie
- verbreiden van de kennis van, het inzicht in, en de belangstelling voor de oncologische gynaecologie
Wij zijn een landelijke multidisciplinaire werkgroep (gynaecologen, radiotherapeuten, medisch oncologen, verpleegkundigen) gericht op alle ontwikkelingen met betrekking tot gynaecologische oncologie. Wij zijn het centraal orgaan en aanspreekpunt in Nederland voor zorgprofessionals binnen de gynaecologische oncologie en signaleren actief knelpunten in de inhoud en organisatie van de zorg, bespreken deze en zetten verbeteracties in gang. Op deze manier werken wij structureel aan het verbeteren van de kwaliteit van de gynaecologische zorg.
De organisatie van de pijler ziet er als volgt uit:
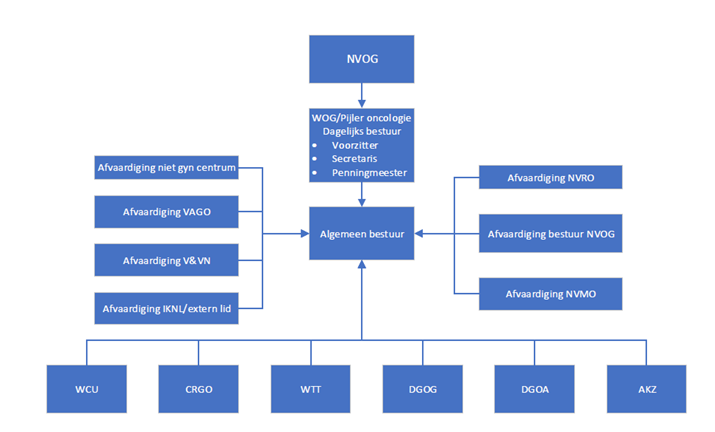
Meer informatie over de activiteiten van iedere werkgroep staat in de rechterkolom onder het kopje Werkgroepen.

